
Investigator Initiated Studies – Research Grant Requests
KARL STORZ is committed to being at the forefront of innovation, advancing scientific and medical knowledge and improving patients’ lives. This commitment is reflected in our robust support of Investigator Initiated Studies (IIS) through research grants.
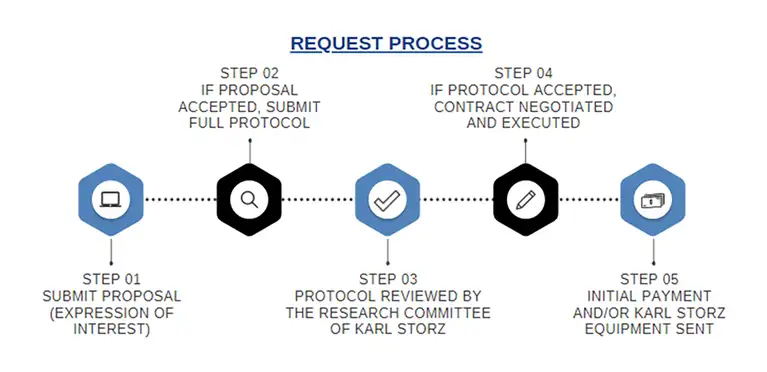
To submit a request for support of an IIS:
Studies Considered for a Research Grant: KARL STORZ considers clinical and pre-clinical research studies in the below “Research Areas of Interest” that are conceived of and implemented by an independent Investigator who assumes the role and responsibilities of the study Sponsor, including ensuring compliance with all regulatory requirements for the duration of the study.
Decision process: Research Grant requests for support of IIS are objectively assessed by the Research Committee of KARL STORZ based on scientific merit and strategic alignment within the “Research Areas of Interest.” In addition, decisions will consider whether the amount of the grant requested is reasonable in consideration of the proposed scope and budget of the research, as well as whether there are sufficient KARL STORZ allocated funds available to support the Research Grant request. Decisions are neither rendered based on KARL STORZ’s past, present or future business opportunities with Investigators nor the Investigator’s incorporation of KARL STORZ products in the research. KARL STORZ reserves the right to deny a Research Grant request in its sole discretion.
Support Provided: If approved, a Research Grant in the form of monetary funding and/or the usage of KARL STORZ products may be provided to support the IIS. Support involving the use of KARL STORZ products is limited to the duration of the IIS and exclusively for purposes of conducting the IIS. All grant support may be subject to public reporting under federal and state transparency laws and regulations, including in Open Payments, in accordance with KARL STORZ policies and applicable legal requirements.
Airway
- Anesthesiology
- Emergency Medicine
Arthroscopy and Sports Medicine /
Arthroscopy General Surgery
- General Surgery
- Mediastinoscopy
- Pediatric Surgery
- Colorectal Surgery
Gynecology
- Hysteroscopy
- Laparoscopy
Urology
- Cystoscopy
- Ureterorenoscopy
- Percutaneous Nephrolithotomy
- Laparoscopy
Integrated Solutions /
OR Integration
- Canary Box
- Neo IP
- AIR
- 4K Boom Room
- AIDA
- StreamConnect
- ComfortOR1
- CollaboratOR
Single Sterile Use and Reusable Endoscopes
Endoscopic Imaging
- Rigid and Flexible Endoscopes
- Light Sources
- Fluorescence
For additional inquiries please contact the Research Team at Research_Grants@karlstorz.com.
** Submission of Proposal and/or Protocol is not a guarantee of funding. The Research Committee of KARL STORZ rigorously vets all submissions, paying particular attention to scientific merit, compliance with policy, and strategic alignment prior to rendering support of any kind.