CORONAVIRUS Pandemic – Our Contribution to the Health Care System
In these turbulent times, we need safety, stability, and particularly the flexibility to quickly adapt to this exceptional situation. The current pandemic presents the health care system, physicians, medical and non-medical staff, patients, and companies with an unprecedented challenge, which can be met only with coordination, communication, and innovation.
In this situation, our focus remains on supporting our partners in health care by providing the necessary products, consultation, and service.
Flexible work – protecting our employees
To ensure that we can continue to serve you, we have taken important steps to protect the safety and health of our employees. We have rigorously implemented social distancing by putting in place the following in-house safeguards throughout the global KARL STORZ Group:
- Staggered work schedules in Production, Logistics, and other areas
- Telecommuting and IT infrastructure expansion to facilitate robust digital collaboration
- Team splitting in critical business areas
- Stricter hygiene and cleaning measures
- A transparent information policy and enhanced employee training
Through these safeguards, we aim to secure our role as your preferred supplier in the health care system so we can continue to make a strong contribution to critical patient care without any disruptions.
Delivery capacity – staying focused
Currently, our Production, Logistics, Service, and all other support functions are fully operational. This is true at our Tuttlingen headquarters and production site as well as at our production sites in Switzerland, Estonia, and the USA. In addition, we are staying in very close contact with our suppliers to ensure our supply of materials. So far, we have not had any critical supply interruptions. We will continue to focus on securing our production capacities to maintain our ability to act in support of patient care. Naturally, the current situation does not lend itself to more detailed forecasts, but thanks to the outstanding commitment of all parties involved, we aim to provide a focused supply at all times. Unfortunately, delays in delivery may already be experienced with certain product groups. In such cases, we remain in close contact with our customers and partners so that they may plan accordingly.
Your contribution is essential
It is very important to us to thank you, as well as the medical and non-medical health care professionals at all health facilities. You work tirelessly to provide the best possible patient care, every day, during this difficult situation. Your commitment serves not only the health of individual patients, but the health of our society as a whole. In this situation, we will do everything in our power to optimally support you in providing patient care. Together, we can make a difference.
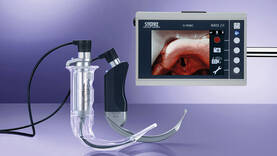
Airway management – video laryngoscopes for intubating Covid-19 patients
In the current Coronavirus situation, the demand for our C-MAC® video laryngoscopes, intubation endoscopes, C-MAC® S single-use video laryngoscopes, and bronchoscopes has considerably risen. To meet this exceptional demand, we have massively increased our manufacturing capacities. We can assure you that we will fully tap all potential resources to continue to considerably and rapidly increase these capacities. The initiated capacity increase is already having a noticeable effect; nevertheless, the current rise in demand can be met only in part, which means that we cannot guarantee immediate delivery at all times.
S-PILOT® – modular smoke evacuation system
As an early pioneer in laparoscopy, KARL STORZ has consistently worked to advance the state of the art. A great example is the S-PILOT® modular smoke evacuation management system, with in-line filtration and options for both active and passive applications. Recent guidance from SAGES and EAES recommends “For MIS procedures, use of devices to filter released CO2 for aerosolized particles should be strongly considered”.
While KARL STORZ has not specifically tested the use of KARL STORZ smoke evacuation devices and SARS-CoV-2, there are data on KARL STORZ filters and its effectiveness against other bacteria and viruses.
The filter efficiency of KARL STORZ filters was tested using the most common test methods with microorganisms known as “Virus Filtration Efficiency (VFE)” and “Bacterial Filtration Efficiency (BFE)”. These types of tests are considered to be the most similar to clinical scenarios. A test aerosol with a mean particle size of 3 +/- 0.3 µm and a virus bacteriophage ɸX174 of 0.027 µm (at VFE) or bacteria Staphylococcus aureus (at BFE) were used. Test results confirmed a filter efficiency of more than 99.9999 %.1,2
The Most Penetrating Particle Size is typically in a range of 0.1-0.3 µm. These particles are the most difficult to trap by the filter. The test aerosol DEHS (= Di-2-Ethylhexyl-Sebacat) with a size of 0.3 µm was therefore used for non-microbiological tests. A filter efficiency of more than 99.9994 % was achieved.3
Contact your local KARL STORZ representative for a solution that meets your individual needs or for more details regarding the smoke evacuation portfolio available.
The S-PILOT® with in-line filtration is part of the extensive array of KARL STORZ products designed in consultation with active laparoscopic surgeons. The complete repertoire covers the full range of diagnostic and surgical laparoscopic procedures for adult and pediatric patients.
1 Nelson Laboratories Inc., Salt Lake City, USA: “Virus Filtration Efficiency Test (VFE) at an Increased Challenge Level” Lab. No. 846021-S01, 07 Dec. 2015 (internal reference: SAP ID 300000338072).
2 Nelson Laboratories Inc., Salt Lake City, USA: “Bacterial Filtration Efficiency Test (BFE) at an Increased Challenge Level” Lab. No. 846022-S01, 02 Dec. 2015 (internal reference: SAP ID 300000338073).
3 GVS Filter Technology UK, Lancashire LA3 3EN, UK: Activity Report REP: 0698/16, 21. January 2016 (internal reference: SAP ID 300000351254).